Chapter 6 periodic table chemistry tes10/31/2022  Squares in the Periodic Table The periodic table displays the symbols and names of the elements, along with information about the structure of their atoms: Atomic number and atomic mass Black symbol = solid red = gas blue = liquid (from the Periodic Table on our classroom wall) Section 6.2 Classifying the Elements OBJECTIVES: Distinguish representative elements and transition metals. Section 6.2 Classifying the Elements OBJECTIVES: Classify elements based on electron configuration. Section 6.2 Classifying the Elements OBJECTIVES: Describe the information in a periodic table. Identified by number & letter (IA, IIA) Īreas of the periodic table Three classes of elements are: 1) metals, 2) nonmetals, and 3) metalloids Metals : electrical conductors, have luster, ductile, malleable Nonmetals : generally brittle and non-lustrous, poor conductors of heat and electricity Īreas of the periodic table Some nonmetals are gases (O, N, Cl) some are brittle solids (S) one is a fuming dark red liquid (Br) Notice the heavy, stair-step line? Metalloids : border the line-2 sides Properties are intermediate between metals and nonmetals Horizontal rows = periods There are 7 periods Vertical column = group (or family) Similar physical & chemical prop. The Periodic Law says: When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. Mendeleev He left blanks for yet undiscovered elements When they were discovered, he had made good predictions But, there were problems: Such as Co and Ni Ar and K Te and I Ī better arrangement In 1913, Henry Moseley – British physicist, arranged elements according to increasing atomic number The arrangement used today The symbol, atomic number & mass are basic items included-textbook page 162 and 163 Īnother possibility: Spiral Periodic Table Mendeleev’s Periodic Table By the mid-1800s, about 70 elements were known to exist Dmitri Mendeleev – a Russian chemist and teacher Arranged elements in order of increasing atomic mass Thus, the first “ Periodic Table ” Dobereiner arranged elements into triads – groups of three elements with similar properties One element in each triad had properties intermediate of the other two elements Section 6.1 Organizing the Elements Chemists used the properties of elements to sort them into groups. As more were discovered, chemists realized they needed a way to organize the elements. Section 6.1 Organizing the Elements A few elements, such as gold and copper, have been known for thousands of years - since ancient times Yet, only about 13 had been identified by the year 1700. Section 6.1 Organizing the Elements OBJECTIVES: Identify three broad classes of elements. 
Section 6.1 Organizing the Elements OBJECTIVES: Compare early and modern periodic tables. Section 6.1 Organizing the Elements OBJECTIVES: Explain how elements are organized in a periodic table. Thank you.Chapter 6 “The Periodic Table” Pre-AP Chemistry Charles Page High School Stephen L. #Chapter 6 periodic table chemistry tes free#Perfect for KS3 Chemistry and lessons can easily be adapted for GCSE Science if required.Įnjoy the resource and feel free to leave a comment. This bundle comes complete with engaging and detailed PowerPoint Presentations, differentiated activities, quizzes, worksheets and class practical activities. *5.1.7 Gas pressure (FREE resource - not included in bundle)*ĥ.4.1 The Periodic Table (metals and non-metals, groups and periods) 
This bundle comes complete with fully differentiated and resourced lessons from the topic. 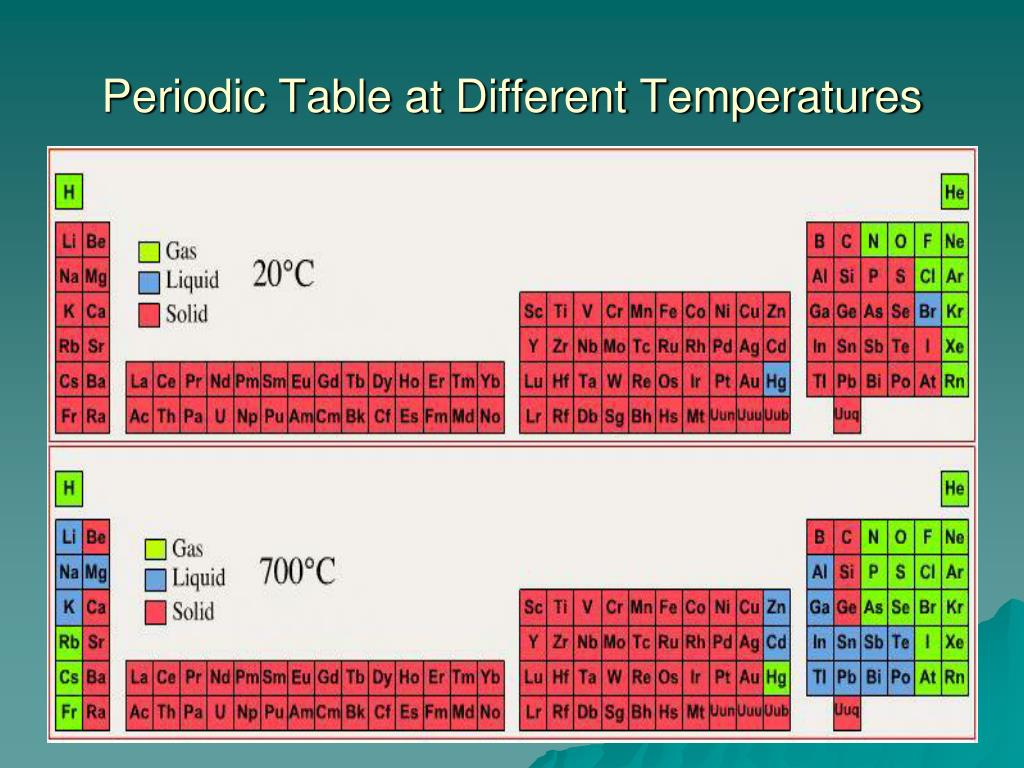
Perfect for KS3 Chemistry! Created for the AQA Activate 1&2 unit 5: Matter. Matter AQA Activate 1&2 KS3 Science Bundle 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
